Abstract
Introduction: Novel agents and autologous stem cell transplant (auto-SCT) have resulted in a survival benefit among patients with multiple myeloma (MM), but almost all ultimately relapse. Allogeneic transplant (allo-SCT) provides a tumor-free graft and graft-versus-myeloma (GVM) effect, but myeloablative conditioning regimens are associated with high rates of treatment-related mortality (TRM). In contrast, reduced intensity allo-SCT conditioning regimens are associated with lower TRM, but patients receiving these regimens tend to have a higher risk of relapse. Radioimmunotherapy (RIT) offers the potential to deliver high doses of targeted radiation while minimizing toxicity to normal tissue. When incorporated into reduced intensity allo-SCT, RIT may improve responses without the associated toxicity historically associated with myeloablative conditioning. We studied 90Yttrium conjugated to an IgG1 murine anti-CD45 monoclonal antibody (MAb) (BC8), in conjunction with a reduced intensity-conditioning regimen prior to allogeneic SCT. We used CD45 as a target due to its ubiquitous expression in hematopoietic stem cells. We used the high-energy (2.8 MeVmax) beta-emitter, 90Y to facilitate a bystander crossfire effect and eliminate malignant plasma cells.
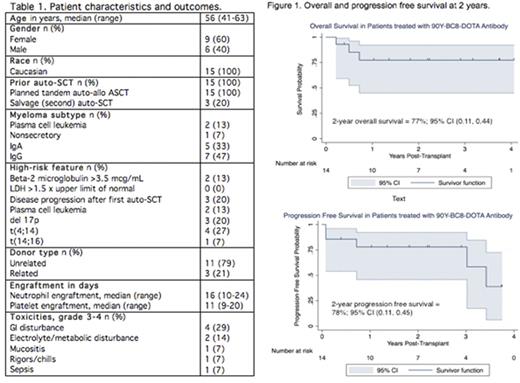
Methods: This is a single-arm, open label, dose-escalation, phase I trial designed to estimate the safety, feasibility and maximum tolerated dose (MTD) of 90Y-BC8-DOTA MAb when combined with fludarabine (flu) and low dose total body irradiation (TBI) followed by HLA‐matched, related or unrelated allo-SCT for patients with high risk MM. Inclusion required patients to have at least one high risk MM-associated disease characteristic at diagnosis (del13 or hypodiploidy by conventional cytogenetics, t(4;14), t(14;16), del 17 by FISH, beta-2 microglobulin >3.5 mcg/mL, LDH >1.5 times upper limit of normal, plasma cell leukemia or progressive disease following primary therapy), and/or disease progression after auto-SCT. To determine the dose of 90Y-BC8-DOTA, patients first underwent a biodistribution step using a trace-labeled infusion of 111Indium-BC8-DOTA followed by gamma camera imaging and bone marrow biopsy. On pre-transplant day -12, patients received 90Y-BC8-DOTA at a dose calculated from the trace-labeled 111Indium-BC8-DOTA biodistribution, followed by Flu (30 mg/m2/day) on days -4 to -2. TBI (2 Gy) was administered on day 0, prior to growth factor mobilized donor peripheral blood stem cell infusion. GVHD prophylaxis consisted of mycophenolate mofetil and cyclosporine initiated on day 0 and day -3, respectively. The MTD was defined as the dose of 90Y-BC8-DOTA MAb associated with a true dose-limiting toxicity (DLT) rate of 25%, where a DLT is defined as Bearman grade III/IV regimen-related toxicity. Doses of 90Y were escalated in increments of 2 Gy depending on the occurrence of DLT.
Results: A total of 15 patients were enrolled. Patient characteristics and outcomes are summarized in Table 1. One patient was withdrawn from the study prior to receiving the therapy dose after testing positive for human anti-mouse antibodies. The remaining 14 patients went on to receive 90Y-BC8-DOTA (0.5 mg/kg antibody) at escalating 90Y dose levels of 6 to 32 Gy to the liver, which is the normal critical organ. Six patients (43%) experienced grade 3-4 toxicity, predominantly GI and metabolic/electrolyte disturbances. No DLTs were observed. Because no DLTs were seen, the MTD could not be estimated. All patients achieved platelet and neutrophil engraftment, and 13 patients (93%) were >95% donor in the CD3, CD33 and CD56 compartments at day +28. Estimates of overall and progression-free survival at 2 years were 77% and 78%, respectively (Figure 1). Eleven patients who completed the study regimen are alive at 2.6 months to 4.1 years. Of these, 6 patients are in complete remission, 2 have persistent disease, and 3 have relapsed at 4 months, 6 months, and 3 years post-transplant. Three patients died: 2 due to progressive disease and 1 due to respiratory failure in the setting of liver GVHD.
Conclusion: The inclusion of 90Y-BC8-DOTA into a reduced intensity allo-SCT conditioning regimen is feasible and no dose-limiting toxicities have been observed. The efficacy of this regimen is promising compared with historical control rates of conventional allo-SCT for MM and should be explored in a phase II trial.
Orozco: Actinium Pharmaceuticals: Other: Research Funding to Institution for sponsored Clinical Trials. Gopal: Seattle Genetics: Consultancy, Research Funding. Becker: GlycoMimetics, Inc.: Research Funding. Press: Roche: Honoraria, Research Funding; BMS: Honoraria; Bayer: Consultancy. Bensinger: Janssen: Membership on an entity's Board of Directors or advisory committees.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal